Contributing writers: Kaliappa Ragunathan and Punit Thakkar, BASF
Corporation
Previously published in CoatingsTech magazine, February 2018.
Architectural exterior coatings are subject to very harsh environmental elements. Extreme conditions like cold and
hot temperatures, low and high humidity, and rain and snow test the ability of coatings to withstand cracking, limit
water absorption, prevent leaching of materials from the surface, and adhere to multiple substrates. Another
detrimental source of damage are UV rays, which induce polymer chain degradation leading to chalking and erosion of
the coatings layer. Surface coatings are also subject to dirt and mildew growth, resulting in a dirty appearance.
When designing a polymer for exterior coatings, inventors must carefully consider these destructive forces.
Professional and do-it-yourself (DIY) painters increasingly care about efficiency — that is, reducing the time
and labor involved in painting surfaces. Paints with paint-and-primer-in-one capabilities help reduce the time and
labor of painting jobs, so developing a polymer that provides both primer and topcoat properties is necessary but
also challenging. This objective further raises the performance requirement for the exterior polymer.
This study focuses on developing a polymer that addresses key needs for paint-and-primer-in-one exterior coatings,
such as dirt pick-up resistance (DPUR), leaching resistance, early rain resistance, gloss retention, grain crack
resistance, adhesion, efflorescence resistance and tannin blocking. The outcome of the study is a polymer that is
designed to withstand the exterior elements in formulations featuring <25 g/l volatile organic compounds (VOC).
Factors Impacting Critical Performance Features
Dirt Pick-Up Resistance (DPUR)
Dirt pick-up is a surface phenomenon and is mainly influenced by hardness of the coating, surface energy, surface
porosity and the nature of the dirt. Softer coating surfaces hold on to dirt better than harder surfaces; therefore,
a harder polymer is required to improve DPUR. Both highly hydrophobic and highly hydrophilic surfaces can provide
better DPUR. However, hydrophobic surfaces are preferred because hydrophilic surfaces reduce water resistance of the
coating. Hydrophobicity can be measured in terms of water contact angle — the higher the contact angle, the
better the hydrophobicity and the higher DPUR. Rough and porous surfaces tend to retain dirt better and consequently
reduce DPUR.
Conflicting Demands: Film Formation at Low VOC and DPUR
A schematic of the film formation process1,2 of emulsion polymer dispersions is depicted in three steps (Figure
1). The steps are 1) water evaporation and particle concentration, 2) particle deformation, and 3) particle
coalescence and polymer chain inter-diffusion. The degree of particle deformation and polymer chain inter-diffusion
depends on the minimum film forming temperature (MFFT) of the particles. Good film formation at low VOC requires
soft polymers or hard polymers with permanent coalescing agents. Permanent coalescing agents remain in the film
forever and reduce the hardness of the coating. Both soft polymers and hard polymers with permanent coalescing
agents lead to reduced DPUR. Thus, low VOC and better DPUR are conflicting demands and a tough problem to solve.
Leaching Resistance

Figure 3: More dirt in leached trail
Rain and dew can leach water-soluble materials from the coatings surface and create a less than desirable appearance
(Figure 2). Leaching resistance of coatings can be improved by reducing water-soluble materials, tailoring film
formation kinetics, improving early water resistance, and incorporating crosslinking chemistry. Leaching of
materials from the surface will ultimately create a porous surface, which can reduce the DPUR of the coatings.
In Figure 3, more red iron oxide adheres to the leached surface, which appears as a darker red trail.
Tannin Blocking and Efflorescence Resistance
Polyphenols (tannin) found in wood and salts in concrete/masonry substrates migrate into coatings and negatively
impact their appearance (Figures 4 and 5). Some of the factors that control these migration phenomena are
film formation kinetics, hydrophobic–hydrophilic balance of the coating, water-soluble materials, functional
groups in the polymer matrix, encapsulation of migrating materials, and adhesion of the coating to the substrate.

Figure 5: Efflorescence on concrete surface
Grain Crack Resistance
Film hardness and elasticity play a critical role in controlling the crack resistance. These two properties need to
be balanced to achieve crack resistance. Another factor that influences crack resistance is emulsion polymer
particle coalescence and film formation. Particle deformation followed by polymer chain inter-diffusion along
particle boundary (Figure 1) provide mechanical integrity to the paint film that is essential for grain crack
resistance. An example where a poorly formulated paint did not provide grain crack resistance in a southern yellow
pine board (red box) is shown in Figure 6. Other paints on the same board provide excellent grain crack
resistance.
Novel Polymer
A novel acrylic polymer was synthesized using the following design principles: particle morphology, low water-soluble
polymers and hydrophobic-hydrophilic balance. Table 1 shows the physical properties of the novel polymer
(Acronal® EDGE 4247). This polymer is free of alkyl phenol ethoxylate surfactants and can
be used to formulate <25 g/l VOC coatings.
Table 1: Novel polymer physical properties
| Property |
Value |
| Weight % solids |
52 to 54 |
| pH |
7.5 to 8.5 |
| Viscosity, cP |
<1000 |
| Particle size (volume average, nm) |
100 to 130 |
| Minimum film forming temperature |
6 to 7 |
The performance of this novel polymer was tested along with four competitive polymers (competitive latex A through
competitive latex D) in an architectural white flat coatings formulation. The formulation and its physical
characteristics are given in Tables 2 and 3, respectively. These paints were tested for topcoat and
primer properties, and the results are discussed in the following section.
Table 2: White flat formulation
| Raw Material |
Pounds |
Gallon |
| Water |
50.0 |
6.0 |
| Titanium dioxide slurry |
294.0 |
15.02 |
| Pigment dispersant |
8.0 |
0.78 |
| Defoamer |
2.0 |
0.28 |
| Wetting agent |
3.0 |
0.35 |
| Biocide |
3.0 |
0.33 |
| Inorganic filler / extender |
230.0 |
10.57 |
| Mineral thixotropic thickener |
4.0 |
0.20 |
| Grind for 20 minutes, then add: |
|
|
| Water |
155.0 |
18.61 |
| Fungicide and an algaecide |
10.0 |
1.03 |
| Neutralizing agent |
1.40 |
0.19 |
| Defoamer |
2.0 |
0.28 |
| Novel polymer (53% weight solids) |
365.0 |
41.25 |
| Polymer particle coalescing agent1 |
6.0 |
0.75 |
| Rheology modifier (high shear) |
35.0 |
4.07 |
| Rheology modifier (low shear) |
2.50 |
0.29 |
| Total |
1170.9 |
100 |
1 – Zero VOC or VOC coalescing agent
Table 3: White flat formulation physical characteristics
| Exterior Flat Formulation |
|
| VOC (gm/L) |
01 or 202 |
| Volume % solids |
41 |
| Weight % solids |
57 |
| PVC |
46 |
1 – Zero VOC coalescing agent
2 – VOC coalescing agent
Experimental
Dirt Pick-up Resistance
Paints were drawn down on a Lenetta black scrub chart using a 250-meter gap bar. The drawn down paints were cured for
24 hours at 72°F and 50 percent humidity. The cured paints were exposed to UV-A radiation for 24 hours in a UV
chamber. Then, either red iron oxide or carbon black dispersion was applied to half of the cured paint. After four
hours, the paints were gently washed with a sponge and running tap water. The washed paints were dried for 24 hours
and the Y reflectance of the soiled and unsoiled areas was measured to calculate DY reflectance.
Surfactant Leaching
Tinted paints (2% Phthalo blue) were drawn down on a Lenetta black scrub chart using a 250-meter gap bar. Tests were
done at cure times of 4 hours and 24 hours. At each cure time, three drops of water were deposited at the top of the
drawn down paints. At 10 minutes, the chart was tilted to run the water off the panel. After 24 hours, the
appearance of the water trails was rated on a 0 to 10 scale with 10 being no change and 0 being severe change.
Efflorescence Resistance
Tiles prepared from plaster of Paris were coated with a specified amount of paint leaving one inch unpainted at one
end. The paints were cured for 24 hours at 72°F and 50 percent humidity. The uncoated ends of the tiles were
placed in a sand bed soaked with 1 percent sodium sulfate solution for two weeks. Then, the appearance of the
painted end of the tiles was recorded.
Early Rain Resistance
Paints were drawn down on aluminum Q-panels using a 250-meter gap bar and dried at 72°F and 50 percent humidity
for 25 minutes. Then, the panels were placed under a 12-inch square shower head and showered for 10 minutes at
constant water flow. The appearance of the test paints was compared to the appearance of a control paint.
Tannin Blocking
Tannin blocking was tested according to ASTM 6686-1 test method. Y reflectance and yellowness index were measured,
not L* and b*.
Corrosion Resistance
Corrosion resistance was tested using cold rolled steel panels according to ASTM G85-11-A5 dilute electrolyte salt
fog test method.
Results and Discussion
Table 4 provides viscosity, film formation rating at 4°C, gloss, contrast ratio, tint strength and scrub
resistance of the coatings. The novel polymer excels in scrub resistance with 1915 scrub cycles at a PVC of 46. This
high scrub resistance indicates excellent film formation and potential for the use of this polymer for even higher
PVC coatings. Tint strength provides indirect evidence for the degree of TiO2 distribution in the coatings
matrix. The novel polymer provides the highest tint strength, while the competitive latices, except competitive
latex B, provide significantly lower tint strength. Higher tint strength indicates lower TiO2 demand to achieve
the same degree of whiteness, resulting in cost savings.
Table 4: Paint Properties
| Property |
Latex A |
Latex B |
Novel Polymer |
Latex C |
Latex D |
| Viscosity (KU) |
108.2 |
113.8 |
106.8 |
105.2 |
98.9 |
| Viscosity (ICI) |
1.6 |
1.9 |
1.4 |
1.8 |
1.6 |
| Film formation1 @ 4°C |
10 |
10 |
10 |
10 |
10 |
| Gloss 20° |
1.5 |
1.5 |
1.5 |
1.5 |
1.5 |
| Gloss 60° |
4.2 |
4.1 |
4.1 |
4.3 |
4.1 |
| Gloss 85° |
3.2 |
3.1 |
3.0 |
3.7 |
4.0 |
| Contrast Ratio |
97.5 |
97.4 |
97.4 |
97.0 |
96.8 |
| Alkyd dry/wet adhesion, 24 hrs |
5B/5B |
5B/5B |
5B/5B |
0/0 |
5B/5B |
| Scrub resistance2 (cycles) |
1655 |
472 |
1915 |
1017 |
353 |
| Tint strength3 |
93.4 |
99.8 |
100 |
93.8 |
92.9 |
1 – 1 to 10 rating scale with 10 being perfect and 1 being
worst
2 – ASTM D 2486 test method
3 – 2% Phthalo blue tint
Dirt Pick-Up Resistance
Two different types of dirt — aqueous iron oxide slurry and aqueous carbon black slurry — were used to
test DPUR performance. The results are shown in Figures 7 and 8. Figure 7 shows DY
reflectance for soiled and unsoiled paint surfaces. The novel polymer has the lowest DY reflectance for both type of
dirt, indicating the best DPUR performance. Figure 8 shows the actual test panels. The novel polymer shows
the best performance from visual examination.
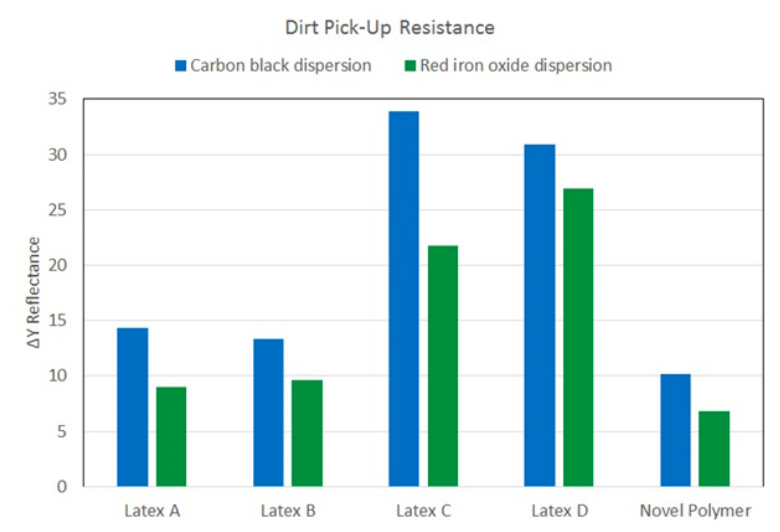
Figure 7: Dirt pick-up resistance

Figure 8: DPUR (carbon black and red iron oxide dispersions)
Surfactant Leaching Resistance
Leaching test results after dry times of 4 hours and 24 hours are shown in Figure 9. The novel polymer and
competitive latex A show similar performance at both dry times, but the other latices show inferior performance.
Early Rain Resistance
Early rain resistance was tested by a shower head water flow test method. The performance at 25 minutes’ dry
time of the novel polymer is compared to a leading 50 g/l VOC commercial paint. As shown in Figure
10, while the novel polymer does not exhibit any defect, the commercial paint does show defect.
Tannin Blocking
Tannin blocking of the coating is tested using Redwood and the results are shown in Figure 11. Figure
11 shows the performance in terms of DY reflectance and D yellowness of the two-coat surface. The novel polymer
has the lowest DY reflectance and D yellowness indicating the lowest amount of tannin migration to the
coating. Figure 12 shows a Redwood board with one and two coats of paint (paint and primer in one
application). The novel polymer with the second coat of the paint has the whitest appearance.
Efflorescence Resistance
The results are shown in Figure 13. The tile coated with the novel polymer-based coating does not show any salt
or defect on the surface, indicating outstanding efflorescence resistance. All other paints show copious amounts of
salt on the surface of the tile due to salt migrating through the coatings layer.
Corrosion Resistance
The novel polymer shows superior corrosion resistance along the scribe and on the surface at 250 hours of testing
(Figure 14).
Paint Film Mechanical Properties
Tensile stress and elongation play a critical role in controlling grain crack resistance. The paint film mechanical
properties for the novel polymer, competitive latex B, and a leading commercial paint are provided in Table 5.
The novel polymer has a much higher elongation without significantly compromising tensile stress. The percent of
elongation is an important feature for withstanding harsh exterior elements, including hot and cold weather and
freeze-thaw cycles.
Table 5: Mechanical properties of white flat paints
| Paint film |
Peak tensile stress (psi) |
% elongation |
| Novel Polymer |
442 |
185 |
| Competitive latex B |
584 |
69 |
| Commercial paint |
745 |
19 |
Accelerated Weathering
A semi-gloss paint formulated with novel polymer and a leading commercial paint were tested for accelerated
weathering according to the ASTM D4587-11 test method using UV-A radiation without condensation cycle. The results
are given in Figure 15 and Table 6. The novel polymer shows excellent gloss retention and minimal
color change.
Table 6: Color change ΔE in accelerated weathering study (UV A radiation)
| Hours |
Novel Polymer |
Commercial Paint |
| 250 |
0.3 |
0.4 |
| 500 |
0.9 |
0.9 |
| 1000 |
1.4 |
1.2 |
| 1500 |
1.3 |
1.3 |
To study the natural weathering of the novel polymer, southern yellow pine and cedar wood boards were coated with
flat paint formulated with the novel polymer, competitive latex 1, competitive latex 2, and a commercial paint. They
were exposed to natural weathering at a 45° angle facing south, in Charlotte, NC. The results at 36 months of
exposure are given in Table 7. The novel polymer is performing very well at 36 months of testing.
Table 7: Natural weathering results at 36 months of testing
| Paint |
Grain Cracking |
Checking |
Flaking |
Chalking* |
Dirt* |
Mildew* |
| Commercial Paint |
None |
None |
None |
9 |
8 |
8 |
| Novel Polymer |
None |
None |
None |
9 |
8 |
8 |
| Latex 1 |
None |
None |
None |
9 |
8 |
8 |
| Latex 2 |
None |
None |
None |
9 |
8 |
8 |
Summary
Architectural exterior coatings are subject to the harsh environmental elements. These harsh conditions must be
carefully considered to overcome the environment’s deleterious effects. The novel polymer developed using
innovative polymer engineering has outstanding performance for paint and primer in one exterior coatings
application. This polymer can be used to formulate <25 g/l VOC coatings. A benchmark study involving market
leading competitive binders shows (Figure 16) that the novel polymer outperforms in DPUR, leaching resistance,
tannin blocking, efflorescence resistance, scrub resistance, corrosion resistance, gloss retention, and tint
strength. Natural weathering shows excellent results at 36 months of testing.
Any questions? Contact a BASF representative to get more information or
request a sample
Get in Touch
Acknowledgments
The authors thank the following for their contributions: Scott Robinson, Shada McCurry, Jeanette Gilliam, Adam
Cummings, Kevin Kearney, Brandi Chew, Julie Adkins, Alan Smith, John Kelly, Kent Clow, Christina Nebel, Sarah
Chronister, Camilo Quiñones-Rozo, Jack Johnson, and Daniel Bates.
References
1. Yoo, J.N., Sperling, L.H., Glinka, C.J., and Klein, A., Macromolecules, 23, 3962 (1990).
2. Taylor, J.W. and Winnik, M.A., “Functional Latex and Thermoset Latex Films,” JCT
Research, (1) 3 163-190 (2004).